เปรียบเทียบกระเป๋า LV Favorite MM Vs PM Damier ของใหม่พร้อมส่งค่ะ
$ 854.50 Buy It Nowor Best Offer, FREE Shipping, 30-Day Returns

Example 13.6 We are given the following atomic masses: 2U = 238.05079 u He = 4.00260 u 90 Th = 234.04363 u H= 1.00783 u 297 Pa = 237.05121 u Here the

If in a group g ,a²b²=b²a²,a³b³=b³a³,then

Louis Vuitton favorite pm vs mm comparison

CHAPTER 10 Electronic Stability Control Systems - ppt video online download

Solved 1. Let u =(3, 1, 2) and v = (1,1,0). a. Find the

LOUIS VUITTON //Favorite pm vs mm

EEEB283 Electrical Machines & Drives - ppt video online download
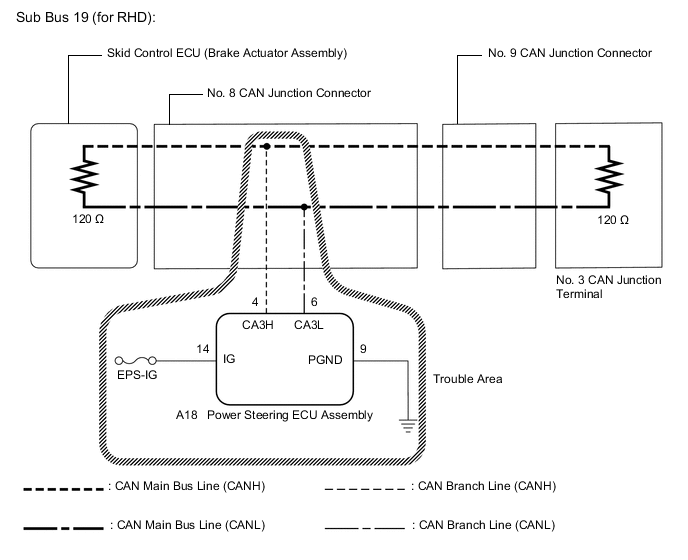
U0131
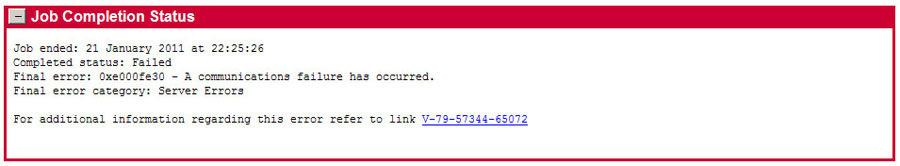
Backup Exec Job Fails With an E000FE30 Error

CM E2 E3 Question Bank, PDF, General Packet Radio Service

LOUIS VUITTON //Favorite pm vs mm
เปรียบเทียบกระเป๋า LV Favorite MM Vs PM Damier ของใหม่พร้อมส่งค่ะ
- Best Price $ 854.50. Good quality and value when compared to berghoff.ir similar items.
- Seller - 509+ items sold. Top-Rated Plus! Top-Rated Seller, 30-day return policy, ships in 1 business day with tracking.
People Also Loved
-

Louis Vuitton Black & White Monogram Gradient Hoodie – Savonches
Buy It Now 3d 15h -

AUTHENTIC Louis Vuitton Multipli Cite GM PREOWNED (WBA346) – Jj's Closet, LLC
Buy It Now 13d 10h -

Coach Mini Bag 616 – TasBatam168
Buy It Now 3d 6h -

Brand New! Louis Vuitton 2054 Heat Reactive Puffer Jacket X Virgil
Buy It Now 6d 13h -

Louis Vuitton White and Black Monogram Silk Scarf - HypedEffect
Buy It Now 23d 9h -

The Gilded Age Antiques & Interiors
Buy It Now 10d 7h -

Repurposed Louis Vuitton Leather Zither Zip Bracelet – N.Kluger
Buy It Now 4d 14h -

Robe Louis Vuitton
Buy It Now 17d 22h -

Sudaderas hombre, prendas de punto para Navidad
Buy It Now 13d 14h -

Louis Vuitton Horizon 55 Suitcase - InteragencyboardShops shop online
Buy It Now 13d 17h -

Numney Men's Wolf Head Leather Belt
Buy It Now 2d 10h -

BaBylissPRO Outlining Trimmer Gold
Buy It Now 17d 10h -

PMguangyin BTS Butter backpack jin jimin suga jungkook V suga bookbag for Gir
Buy It Now 13d 13h -

Louis Vuitton Damier Ebene Favorite PM QJB0YV0T0F045
Buy It Now 7d 9h -

The Chanel Gabrielle Bag Has Proved to Be The Brand's Latest in a
Buy It Now 17d 11h -
Kardashian Kids Now Own Louis Vuitton Bags
Buy It Now 19d 17h -

Gucci Beige GG Canvas Pochette Bag
Buy It Now 4d 23h -

Louis Vuitton guide
Buy It Now 11d 15h -

NBA playoffs 2022: Eye-catching outfits from the first round of the postseason
Buy It Now 8d 23h -

Venus Knitted Backpack - Made in Taiwan - Bucket Bag - Knitted Bag
Buy It Now 18d 5h -

FOR IPHONE 15 PRO MAX (6.7) SIMPLEMADE MAGSAFE COMPATIBLE LIQUID
Buy It Now 8d 10h -

Gifts Idea Thelma Movie Fim Louise Gifts For Birthday Poster for Sale by GaudenBozzelli
Buy It Now 19d 21h -

Louis Vuitton Blue Taurillon Leather Slender Wallet M80590 w/ Receipt LV
Buy It Now 2d 18h -

Gucci Savoy large duffle bag
Buy It Now 5d 13h
